Autonomic Dysreflexia
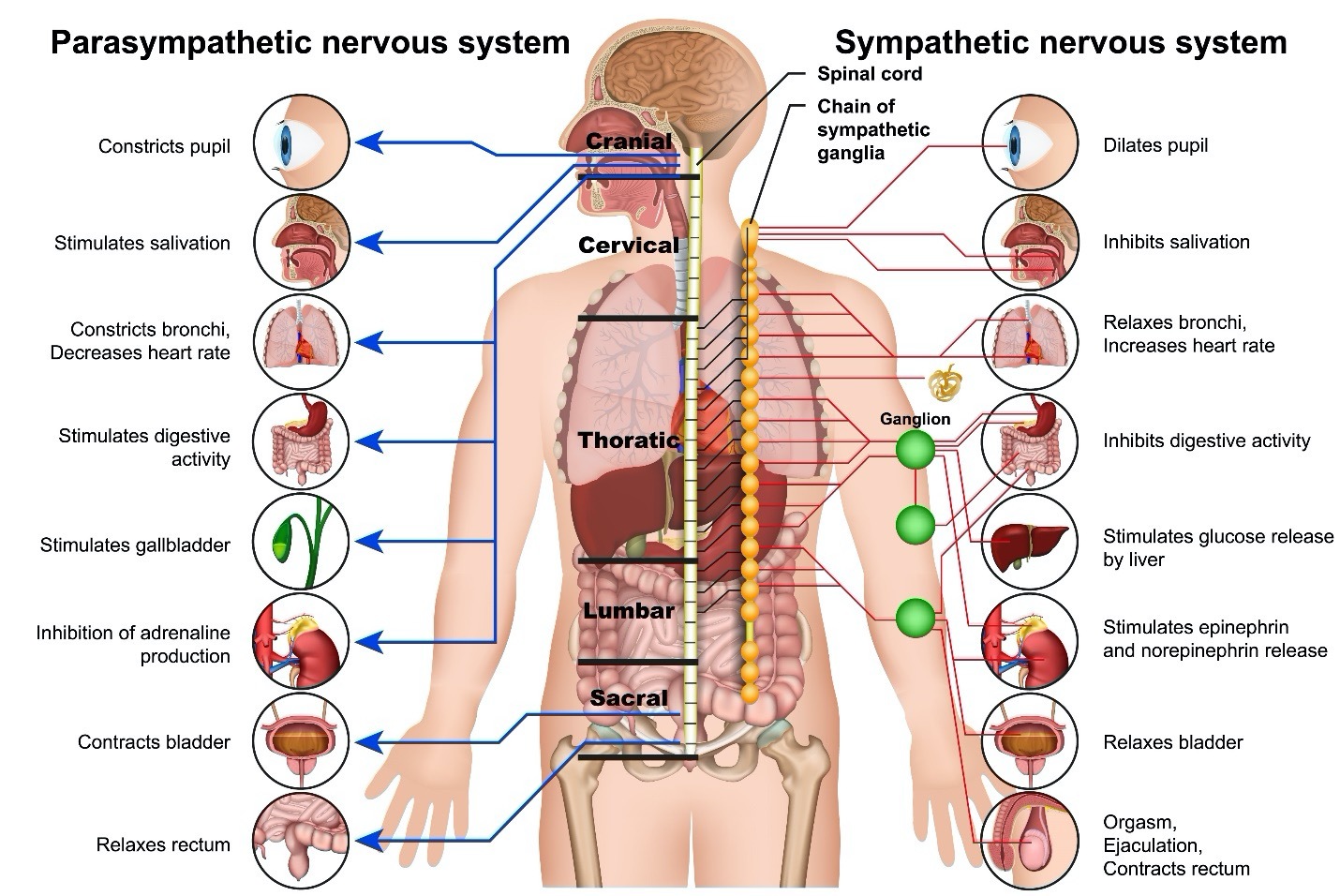
The nervous system is a complicated network composed of many parts and subsets. One of these parts is the autonomic nervous system (ANS) which controls all body functions that you do not think about to make occur. The ANS balances its subparts, the sympathetic and parasympathetic nervous system. The sympathetic autonomic nervous system (SANS) speeds up body reactions while the parasympathetic autonomic nervous system (PANS) slows these reactions. This combination keeps the body functioning in harmony. When threatened, the sympathetic nervous system speeds without containment by the parasympathetic system which results in the fight or flight response. If the body is threatened, a release of energy bursts through the nervous system as a reaction. This is the usual human response to a threat.
Comparisons of some of the functions of the Sympathetic and Parasympathetic Nervous Systems:
Autonomic dysreflexia (AD) is an issue in the autonomic nervous system. Historical terms for AD that may still be heard today are dysautonomia and hyperreflexia because there is too much (hyper) reflexic response. The autonomic nervous system (ANS) is the part of the nervous system that controls activity in the body without your consciously thinking about it. This is what keeps the body functioning so you can concentrate on other things.
Anything that occurs within your body that happens automatically or not under your control is managed by the autonomic nervous system. These body activities include hundreds of activities such as your heart beating, breathing, digestion, blood pressure regulation, body temperature control, response to infection, sweating, itching, pain control, keeping your muscles relaxed, sexual arousal and the list goes on. Every function of your body that works without conscious thought is controlled by the autonomic nervous system.
Without injury to the nervous system, the body senses noxious or unpleasant stimulations through the sensory nerves in the body. These nerves then send a signal to the brain about what the issue is and specifics about the location. The brain interprets the signal and sends an appropriate response through the motor nerves to move, turn, scratch, adjust the temperature of the body, control blood pressure, or in some way adjust the body.
Motor nerves control all the movement in the body. Movement includes moving your muscles to allow function to your arms and legs but also to adjust temperature and blood pressure as well as internal body functioning. Located in the brain are upper motor neurons (nerve cells) with their axon or arm mostly reaching into the higher spinal cord. Interneurons connect upper and lower motor neurons within the spinal cord. The spinal cord is the location of lower motor neurons (nerves) with their axon or arm leaving the spinal canal and extending into the body itself.
Injury that affects upper motor neurons produces autonomic dysreflexia (AD). Therefore, individuals with spinal cord injury in the cervical or thoracic regions of the spinal cord (above T6) are at risk for AD because this is the location of the upper motor neuron axons or arms. Also at risk are individuals with brain injury, stroke, and other neurological diseases if the upper motor neurons in the brain or upper spinal cord are affected.
After a nervous system injury, communications to and from the brain may be partial, absent, or misinterpreted messages. However, messages are still being sent and received through the nerves of the ANS. Nerves are throughout the body, including the area of injury. Your automatic nervous system is still working but perhaps not at the same level as before your injury. Instead of a small response to just a specific issue, an energy burst from the sympathetic nervous system is released as a radical correction. The parasympathetic nervous system is unable to slow the release. This is autonomic dysreflexia, an automatic reflex over-response that cannot be contained. Because the ANS is automatically controlled, you cannot consciously change or control your body’s autonomic dysreflexia response. Emergency treatment should be started immediately.
Who is at Risk for Autonomic Dysreflexia
Individuals with injury from disease or trauma to the upper motor nerves are at risk for autonomic dysreflexia. Traditionally, the presence of a spinal cord injury above T6 is the determining factor for AD but recent research indicates those with injury at T10 and above have reported symptoms.
Individuals with other injuries affecting the upper motor neurons are also at risk for AD. These include individuals with a diagnosis of brain injury, stroke, multiple sclerosis, Guillain-Barré syndrome, or other neurological diseases. Any disease or injury affecting the autonomic nervous system can result in episodes of autonomic dysreflexia. A particular type of autonomic disruption after brain injury is paroxysmal sympathetic hyperactivity (PSH) which has the symptoms of transient fever, tachycardia (rapid heart rate), hypertension (high blood pressure), tachypnea (rapid breathing), excessive diaphoresis (sweating), and specific posturing.
Being diagnosed with any of the above listed neurological conditions, does not mean you will develop AD. It is not understood why some individuals develop AD while others do not. Nor is it completely understood why some have more severe symptoms than others. There are some rare cases of individuals who have AD without a neurological diagnosis or without knowing the specific cause.
What are the Symptoms of Autonomic Dysreflexia (AD)
The characteristics of symptoms for AD vary by individual. Some individuals have extremely mild symptoms that are unfortunately often ignored. Others have incapacitating symptoms. Please note any, a few, or all the symptoms can be experienced with AD. Some symptoms will appear with different triggers. Most individuals learn about the pounding headache as a symptom of AD. A pounding headache is the most often cited symptom but any of these symptoms listed can be noted even without the pounding headache. Ignoring any symptom can be life threatening, even if mild. Blood pressure monitoring is essential in detecting AD. Elevated blood pressure above your normal range can lead to stroke, cardiac arrest, seizures, retinal hemorrhage, pulmonary edema and even death if untreated.
Typical symptoms of AD in adults are:
ABOVE LEVEL OF INJURY
- Hypertension (A fast increase in blood pressure, 20-40 mm Hg systolic higher than usual)
- Bradycardia (slow heart rate) or Tachycardia (fast heart rate)
- Pounding headache
- Apprehension/anxiety/ uneasy feeling
- Changes in vision
- Nasal congestion
- Sweating
- Flushed skin
- Goosebumps
- Tingling sensation
BELOW LEVEL OF INJURY
- Nausea
- Chills without fever
- Clammy
- Cool
- Pale
Typical symptoms of AD in children are:
ABOVE LEVEL OF INJURY
- Hypertension (A fast increase in blood pressure, 15 mm Hg systolic higher than usual in children and 15-20 mm Hg systolic higher than usual in adolescents)
- Bradycardia (slow heart rate) or Tachycardia (fast heart rate)
- Big headache
- Feeling nervous/worried/scared
- Red cheeks/neck/shoulders
- Blurry vision
- Stuffy nose
- Sweating
- Goosebumps
- Tingling
BELOW LEVEL OF INJURY
- Upset stomach, feels like you need to throw up
- Chills without fever
- Clammy or cold and sweaty
- Cool
- Pale
What is Silent Autonomic Dysreflexia
More evidence is being discovered that some individuals have AD episodes without symptoms. In other words, elevated blood pressure without any alerts or symptoms in the body. These discoveries have been observed when blood pressure is monitored during sleep, in bladder assessment procedures, and during routine bowel programs. In all these cases, blood pressure is at AD diagnosis/elevation level, but the individual has no symptoms. It is important that during routine activities, your blood pressure is monitored to assess for AD. If there is no change in your blood pressure, occasional monitoring can be done. If there are changes in your blood pressure, immediately notify your healthcare professional for further evaluation and instruction.
How Autonomic Dysreflexia is Diagnosed
Detecting the presence of AD is performed through assessment of blood pressure. Most often a symptom or multiple symptoms are noted at which time, blood pressure should be measured. Individuals need to know their usual blood pressure as over time with neurological conditions, especially spinal cord injury, blood pressure tends to lower due to cardiac changes through slowed blood vessel response. Adults with spinal cord injury at T6 or above may not retain their average pre-injury blood pressure but often will have a blood pressure that is normal for them at 90-110mmg Hg systolic (top number). You need to know your normal blood pressure to recognize an increase in adults of 20-40mmg Hg systolic (the top number), in teens an increase of 15-20mmg Hg systolic, and in children an increase of 15mmg Hg systolic.
Autonomic dysreflexia will be suspected in individuals with injury to upper motor neurons. The onset of an episode of AD can occur at any time in individuals with neurologic disease or injury such as stroke, brain injury, multiple sclerosis, Guillain-Barré syndrome, and others. Some neurological diseases are progressive so onset time cannot be determined as it occurs when an individual reaches an unknown point in the disease.
For those with injury to the spinal cord in the cervical or thoracic region, AD classically occurs in injury above T6 due to the effects of a bundle of nerves at T5. Some individuals with injury at T10 and above have noticed AD because of extended trauma and the uniqueness of an individual’s anatomy. AD was thought not to occur until spinal shock has resolved which is typically about six weeks from trauma onset but can last longer. However, recent studies indicate AD can occur within the first month of injury. Those with spinal cord injury in the lumbar or sacral area have injury in the lower motor nerves which does not produce AD.
There is no blood test or radiological process that can detect AD however catecholamines and vasopressin levels in blood can be elevated during or for a time after an AD episode.
What are Triggers for Autonomic Dysreflexia
The detection of AD has been classically noted to be due to a trigger that stimulates the body into an AD episode. A trigger is something that the body perceives as noxious or irritating below the level of injury. If the message that something small or big is bothering the body where sensation is decreased, the sensory nerves cannot effectively or efficiently send a message to the brain to correct the situation. As a result, the ANS would typically send a message to the motor nerves to move the body which would correct the situation. However, due to nerve miscommunication, a total blast of messages is sent. The messages are unable to get through which results in this massive burst of nerve activity in the body, usually above the level of injury.
With the discovery of silent AD, triggers may not be noted or act as a stimulus for an AD episode. In silent AD, blood pressure is affected without the identification of a trigger. An AD episode may occur without symptoms which is why it is called silent AD.
Triggers include a variety of noxious sources or irritations to the body. Common triggers are listed below. Individuals have different triggers. The most common noxious irritations that stimulate episodes of AD are from the bladder, bowel, and skin. However, the list of possible triggers is always increasing. Triggers fit into categories, although your trigger may be quite unique.
- The bladder is the primary documented source of triggering AD episodes. Most often the cause is something that is not allowing urine to pass out of the body leading to bladder overdistention. This can range from a kink in a catheter, clogged catheter, bladder spasms, detrusor sphincter dyssynergia (DSD, the bladder and sphincter not working in unison), stones, infection, or other constrictions. For some, intermittent catheterization or suprapubic catheters can be an irritant. Bladder testing through cystoscopy (a scope inserted into the bladder), urodynamic testing (assessment of bladder function), shock-wave lithotripsy (breaking up stones) can also trigger AD. Equipment that is used for urine containment such as leg bag straps that are too tight or a heavy or overfull leg bag, wet urine containment undergarments, or external catheter straps are also culprits.
- The second most frequent trigger of AD is the bowel. Bowel overdistention from swift movement, an incomplete or lack of a bowel program, impaction, constipation, gas, regular or overzealous digital stimulation, enemas, or manual removal are all triggers. Diarrhea or internal issues such as diverticulitis, Crohn’s disease, fissures, and hemorrhoids can also be noxious irritants.
- Skin issues are the third most common trigger of AD. Skin issues cover a wide range from an itch that cannot be sensed or scratched, rashes, pressure injury at any stage, cuts, bruises, bone fractures, ingrown toenails, overly long toenails, sitting on a wrinkled sheet, even just an air current blowing over the fine hairs of your arm or leg. Constrictive clothing that is too tight, lumpy, or baggy, rivets on jeans, shoes that fit incorrectly, socks too tight in the toes or leg, belts, bras, the list of clothing issues is constantly increasing.
As more triggers for AD are being identified, it seems anything bothersome can be an issue. These are some additional triggers:
- Temperature changes internally such as fever or chills, or externally such as a warm or cold environment
- Spasticity anywhere in the body
- Internal issues such as gallbladder attacks/stones, appendicitis, liver function issues, pancreatitis, ovarian cyst, prostatitis, ulcers in the digestive tract, gastric reflux, etc.
- Sexual function and interaction, menstruation, pregnancy, delivery, sperm retrieval
- Deep vein thrombosis (DVT) (formation of a blood clot in a deep vein), pulmonary embolism (PE)
- Heterotrophic ossification (HO) (presence of bone in soft tissue where it is not usually found)
- Orthostatic hypotension (OH) (blood pressure drops when sitting or standing)
- Pain
- Eye effects from computer use or bright sunshine
What is the Treatment for Autonomic Dysreflexia
AD is a medical emergency. When blood pressure is raised, you need to act immediately. Do not hesitate to call 911 if needed.
- When AD symptoms are noted, start by first quickly sitting bolt upright. Your torso and hips should be at a 90-degree angle. Have someone help you to a sitting position if you are unable to do this yourself. The sudden change from laying to sitting takes advantage of orthostatic hypotension when your blood pressure suddenly drops as the blood vessels cannot constrict to rush blood to your head fast enough.
- Continue to monitor blood pressure every 2-3 minutes until it returns to your normal.
- Loosen anything tight or restrictive on your body while getting into the sitting position.
- Look for the cause of this episode of AD. Start with checking the three most common triggers for AD. Check urine flow. Catheterize if necessary (if there is no or little urine output). Then check the bowel for blockage. Disimpact the bowel if stool is present. Then check the skin removing wrinkles, constrictions or tight clothes. You may know your triggers from previous AD episodes. Correction of your usual trigger source is a good start if you are aware of it. If AD does not start to resolve with corrections, continue to look for and remove triggers below the level of injury.
- If you have medication prescribed for AD administer it. Medication may consist of an antihypertensive with rapid onset and short duration. Commonly prescribed medications are:
-
-
- Nitro Paste—1/2” (for individuals younger than 13 years) or 1” (for individuals 13 years and older), apply every 30 minutes, topically above level of injury. Reapply if needed. Wash off the Nitro paste when blood pressure is stable to avoid lowering blood pressure too much.
- Nifedipine (if Nitro paste is NOT available)– 0.25-0.5mg/kg per dose (for individuals younger than 13 years old) or 10mg per dose (for individuals 13 years and older), squirt immediate release form sublingually (under your tongue) or if in pill form, chew it. This may be repeated every 20-30 min as needed.
- IV antihypertensives may be administered only in a monitored setting (I.C.U.) as close monitoring is required.
- Other medications used to treat ongoing and dangerous AD episodes include nifedipine, nitrates, captopril, terazosin, prazosin, phenoxybenzamine, prostaglandin ED and sildenafil. Selection of the correct medication for you should be made in consultation with your healthcare professional.
-
6.Your blood pressure should begin to lower and correct itself. Continue to monitor your blood pressure for at least two hours.
7.If the trigger or multiple triggers are found and removed or corrected but blood pressure remains elevated, call 911. Medical attention is needed immediately to prevent a cardiovascular event such as stroke, cardiac arrest, seizures, retinal hemorrhage, pulmonary edema, and death.
If you have identified specific triggers for AD, other medications and treatments can be used to help control those known causes. This might include medication for detrusor sphincter dyssynergia (DSD), sphincterotomy (a small enlargement in a male’s urinary sphincter to resolve increased bladder pressure), change of type of catheter used, or adding numbing medication during catheter insertion.
Alterations in diet for stool consistency, use of a gentle approach to suppository and digital stimulation, or local anesthetic for bowel programs will help if bowel issues are AD triggers.
To aid in reduction of skin triggers, medication can be used to control tone (spasms), ensure you are not sitting on a wrinkle or other skin irritant, perform pressure releases and check your skin for pressure injury, as well as use of pressure dispersing equipment, and monitor your leg bag if you use one to ensure it is not pulling on your leg as it fills with urine, check your shoes to be sure they fit and maintain foot hygiene avoiding cracked callouses and ingrown toenails. Be cautious with your body when moving to avoid injury. Protect your eyes from computer overuse and bright sunshine.
Autonomic Dysreflexia Video
A video explaining the cause and treatments for Autonomic Dysreflexia from the Reeve Foundation can be viewed here.
Autonomic Dysreflexia Wallet Card
To ensure you have up to date information about Autonomic Dysreflexia (AD), download the Christopher & Dana Reeve Foundation National Paralysis Resource Center’s Autonomic Dysreflexia Wallet Card. This card can be shared with healthcare professionals, caregivers, teachers, and others. The wallet card is an easy to carry trifold card that can be placed in your wallet for quick access. It is available for adults and children as well as in a variety of languages. Download the AD wallet card from: https://www.ChristopherReeve.org/cards or call the Reeve Information Specialists for a free print copy at 800-539-7309.
Rehabilitation for Autonomic Dysreflexia
You will need to be involved with identification of AD episodes. The moment you start to feel an episode of AD is occurring, begin treatment by sitting up. Call for assistance to be sure you do not fall, harm yourself, or lose consciousness. Someone should call 911 if the trigger cannot be identified or if treatment resolutions are not working.
A physiatrist, neurologist, primary care physician, medical doctor, or nurse practitioner that can diagnose AD will provide treatments that are appropriate for your AD episodes. If the episodes are easily corrected, monitoring with careful consideration of avoiding identified triggers may be necessary. In more severe cases, medications to treat AD will be prescribed. Also, if your trigger is known, medication can be prescribed to reduce the trigger thereby reducing or eliminating AD episodes.
A Rehabilitation Registered Nurse is a care provider in the hospital, rehabilitation hospital or community that can educate you about AD, help you learn to identify it, and how to treat it. They will help you learn to deal with emergency situations.
A Urologist may provide treatment such as bladder numbing agents and treatments to avoid triggering AD.
Care providers at home can assist with monitoring you for AD episodes as well as providing emergency care. They are typically the person to call 911, as necessary.
If you have AD, you need to let all your healthcare providers and caregivers know about your condition in case it arises, and you do not have time to call for help. This includes therapists who will provide therapy using strategies to avoid an AD episode and all caregivers who can ensure your safety.
Research
Traditionally, research about causes and treatments for autonomic dysreflexia has focused on the individual trigger such as tone (spasms), pressure, bladder, and bowel issues. However, healthcare professionals note those individuals who are treated for tone or neuropathic pain have less incidence of AD. This has sparked an interest in examining the autonomic nervous system as a whole for treatments rather than just one subset of causes (Rabchevsky, et al., 2011).
A study of autonomic nervous system tests was completed to see if predictors for AD could be isolated. Deep breathing, Valsalva maneuver and tilt table test challenged individuals with spinal cord injury. Measurements of baroreflex sensitivity (BRS), and spectral analysis of heart rate and blood pressure variability were assessed along with blood level of catecholamines and vasopressin levels and clinical and radiological examinations. AD was demonstrated by 73.3% of SCI patients, 63.6% of those were asymptomatic (Cívicos Sánchez, et al., 2021). Although this study is not practical in clinical use at this time, it is an effective assessment of those who have AD perhaps identifying those without symptoms.
Silent AD has been recently brought to the forefront of care for individuals with ANS disruption. Previously, AD was thought to be only in individuals with the manifestation of symptoms. Recently scientists discovered AD in individuals who did not have any symptoms. This is exhibited by elevated blood pressure but no changes in body comfort. Silent AD is now a critical issue for individuals who are at risk of serious complications but have no warning (Kirshblum, et al., 2002; Linsenmeyer, et al., 1996.)
Research in mice demonstrates that AD affects further decreases in function in the immune system. A follow up study of one individual noted an excessive catecholamine release affecting the immune system which indicates that AD can be a secondary cause of immune deficiency after SCI (Zhang, et al., 2013). This new information will require further study as SCI has an effect on decreased immune functioning. Added with neuroinflammation of the nervous system, further effects of AD on the immune system are being studied with treatments yet to be defined (Mironets, et al., 2018)
Facts and Figures
Individuals with spinal cord injury at the T6 level or above have a rate of AD of 48-90%.
Silent AD is prevalent in individuals with SCI at the T6 level or above at a rate of 42.9-63.6%.
The rate of AD in children 13 years and younger with spinal cord injury at T6 or above in a single site study was 51%.
AD mortality rate is 22% and increases the risk of stroke by 300% to 400%.
Autonomic Dysreflexia occurs more often in individuals with complete spinal cord classification at 91%, than with incomplete classification at 27%. Time of onset of AD is shorter for those with complete spinal cord classification and longer for those with incomplete classification.
Incidence of autonomic dysfunction in brain trauma in one study of 349 reported cases:
- Traumatic Brain Injury (TBI) 79.4%
- Hypoxia 9.7%
- Cerebrovascular accident or stroke 5.4%
Consumer Resources
If you are looking for more information about spinal cord injury or have a specific question, our Information Specialists are available business weekdays, Monday through Friday, toll-free at 800-539-7309 from 9:00 am to 8:00 pm ET.
Additionally, the Reeve Foundation’s National Paralysis Resource Center maintains an autonomic dysreflexia fact sheet with additional resources from trusted Reeve Foundation sources. Reeve also offers a fact sheet about the autonomic nervous system. Check out our repository of fact sheets on hundreds of topics ranging from state resources to secondary complications of paralysis.
We encourage you to reach out to organizations, including associations which feature news, research support, and resources as well as a national network of support groups, clinics, and specialty hospitals.
Clinical Guidelines:
Consortium for Spinal Cord Injury Medicine. Acute Management of Autonomic Dysreflexia: Individuals with Spinal Cord Injury Presenting to Health-Care Facilities 2nd Edition. © Copyright 2001, Paralyzed Veterans of America. https://pva-cdnendpoint.azureedge.net/prod/libraries/media/pva/library/publications/cpg_autonomic-dysreflexia.pdf
Washington State Department of Health, Office of Community Health Systems Emergency Medical Services & Trauma Section Trauma Rehabilitation Clinical Guideline Autonomic Dysreflexia, January 15, 2020. https://www.doh.wa.gov/Portals/1/Documents/Pubs/530237.pdf
References
Baguley IJ, Perkes IE, Fernandez-Ortega JF, Rabinstein AA, Dolce G, Hendricks HT; Consensus Working Group. Paroxysmal sympathetic hyperactivity after acquired brain injury: consensus on conceptual definition, nomenclature, and diagnostic criteria. J Neurotrauma. 2014 Sep 1;31(17):1515-20. doi: 10.1089/neu.2013.3301. Epub 2014 Jul 28. PMID: 24731076.
Canon S, Shera A, Phan NM, Lapicz L, Scheidweiler T, Batchelor L, Swearingen C. Autonomic dysreflexia during urodynamics in children and adolescents with spinal cord injury or severe neurologic disease. J Pediatr Urol. 2015 Feb;11(1):32.e1-4. doi: 10.1016/j.jpurol.2014.08.011. Epub 2014 Oct 8. PMID: 25697979.
Caruso D, Gater D, Harnish C. Prevention of recurrent autonomic dysreflexia: a survey of current practice. Clin Auton Res. 2015 Oct;25(5):293-300. doi: 10.1007/s10286-015-0303-0. Epub 2015 Aug 18. PMID: 26280219.
Cívicos Sánchez N, Acera M, Murueta-Goyena A, Sagastibeltza N, Martínez R, Cuadrado M, Orueta A, Tijero B, Fernández T, Del Pino R, Gabilondo I, Jauregui Abrisqueta ML, Gómez Esteban JC. Quantitative analysis of dysautonomia in patients with autonomic dysreflexia. J Neurol. 2021 Aug;268(8):2985-2994. doi: 10.1007/s00415-021-10478-w. Epub 2021 Feb 25. PMID: 33634338.
Cowan H, Lakra C, Desai M, Autonomic dysreflexia in spinal cord injury. BMJ 2020;371:m3596. doi: https://doi.org/10.1136/bmj.m3596.
Ekland MB, Krassioukov AV, McBride KE, Elliott SL. Incidence of autonomic dysreflexia and silent autonomic dysreflexia in men with spinal cord injury undergoing sperm retrieval: implications for clinical practice. J Spinal Cord Med. 2008;31(1):33-9. doi: 10.1080/10790268.2008.11753978. PMID: 18533409; PMCID: PMC2435024.
Eldahan KC, Rabchevsky AG. Autonomic dysreflexia after spinal cord injury: Systemic pathophysiology and methods of management. Auton Neurosci. 2018 Jan;209:59-70. doi: 10.1016/j.autneu.2017.05.002. Epub 2017 May 8. PMID: 28506502; PMCID: PMC5677594.
Giannantoni A, Di Stasi SM, Scivoletto G, Mollo A, Silecchia A, Fuoco U, Vespasiani G. Autonomic dysreflexia during urodynamics. Spinal Cord. 1998 Nov;36(11):756-60. doi: 10.1038/sj.sc.3100684. PMID: 9848482.
Helkowski WM, Ditunno JF Jr, Boninger M. Autonomic dysreflexia: incidence in persons with neurologically complete and incomplete tetraplegia. J Spinal Cord Med. 2003 Fall;26(3):244-7. doi: 10.1080/10790268.2003.11753691. PMID: 14997966.
Hickey KJ, Vogel LC, Willis KM, Anderson CJ. Prevalence and etiology of autonomic dysreflexia in children with spinal cord injuries. J Spinal Cord Med. 2004;27 Suppl 1:S54-60. doi: 10.1080/10790268.2004.11753786. PMID: 15503704.
Huang YH, Bih LI, Liao JM, Chen SL, Chou LW, Lin PH. Blood pressure and age associated with silent autonomic dysreflexia during urodynamic examinations in patients with spinal cord injury. Spinal Cord. 2013 May;51(5):401-5. doi: 10.1038/sc.2012.155. Epub 2012 Dec 11. PMID: 23229618.
Karlsson AK. Autonomic dysreflexia. Spinal Cord. 1999 Jun;37(6):383-91. doi: 10.1038/sj.sc.3100867. PMID: 10432257.
Kirshblum SC, House JG, O’connor KC. Silent autonomic dysreflexia during a routine bowel program in persons with traumatic spinal cord injury: a preliminary study. Arch Phys Med Rehabil. 2002 Dec;83(12):1774-6. doi: 10.1053/apmr.2002.36070. PMID: 12474185.
Krassioukov AV, Furlan JC, Fehlings MG. Autonomic dysreflexia in acut